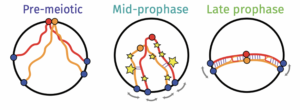
Meiosis is a specialized cell division that is required for the formation of haploid gametes and is essential for sexual reproduction. We are interested in the unique chromosome events that prepare homologous chromosomes to separate from one another at meiosis I.
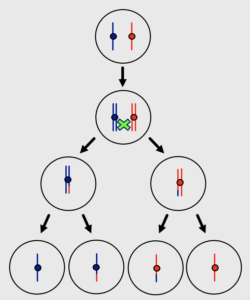
Prophase I is the stage of meiosis when homologous chromosomes undergo pairing, recombination and synapsis of homologous chromosomes . Understanding the mechanisms underlying the dynamic chromosomal events of meiosis can help us to understand how genomes are passed from one generation to the next.
We use both budding yeast Saccharomyces cerevisiae and the zebrafish Danio rerio as model organisms to study these events. We collaborate with Dr. Andrew Spakowitz at Stanford University to describe chromosome movements using polymer theory and Dr. Bruce Draper, Department of Molecular and Cellular Biology to study zebrafish gametogenesis
Research topics:
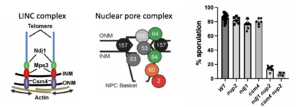
1. Interplay between the LINC complex and the Nuclear Pore Complex in yeast
Telomeres are attached to actin filaments outside the nucleus via the LINC complex that direct rapid prophase movements. Yeast proteins involved in this process include Ndj1 and Csm4. We found that deleting NUP2 or NUP60 nucleoporins will have a synthetic sporulation defect when combined with mutations in either NDJ1 or CSM4 suggesting that the LINKC complex and the NPC are two levels of meiotic chromosome organization. We recently found that a fragment of Nup2 relieves an inhibition of Nup60 to promote meiosis and sporulation. You can read more about this work here: Komachi and Burgess, 2022
2. Diffusion and distal linkages govern inter-chromosomal dynamics during meiotic prophase in yeast
In collaboration with our colleague Andrew Spakowitz at Stanford University, we found that the interactions between homologous chromosomes is more dynamic than anticipated. We also showed that the measured relative changes in distance between tagged homologous loci can be modeled using modified viscoelastic Rouse polymer model. You can read more about this work here: Newman et al 2022
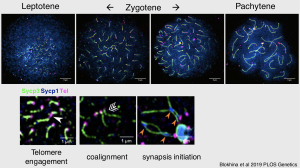
3. The telomere bouquet is a hub for meiotic chromosome dynamics in zebrafish
We use super-resolution microscopy to observe chromosome pairing behavior in zebrafish. The clustering of telomeres in the bouquet provides a space in which the ends of chromosomes coalign and synapse by the formation of the synaptonemal complex. You can read more about this work here: Blokhina et al 2019